Azimuthal Quantum Number
Azimuthal Quantum Number Assignment Help | Azimuthal Quantum Number Homework Help
Azimuthal Quantum Number
It refers to the subshell or sublevel to which an electron belongs and describes the motion of the electron. The magnitude of the angular momentum of an electron (angular velocity x moment of inertia) is related to by the expression:
______________________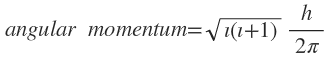
where =0,1,2,3,........,(n-1).
This means that the values of for the subshells of a shell are govermedby n. When n=1, there is only one subshell with the value . When n=2 there are two subshells with the values =0. 1. when n=3 there are three subshells that have values =0,1,2. Similarly, when n=4.. no. of subshells=4 with values = 0,1,2,3. For every value of . there is a separate notation to indicate subshell. For example.
______________________
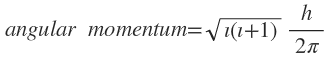
where =0,1,2,3,........,(n-1).
This means that the values of for the subshells of a shell are govermedby n. When n=1, there is only one subshell with the value . When n=2 there are two subshells with the values =0. 1. when n=3 there are three subshells that have values =0,1,2. Similarly, when n=4.. no. of subshells=4 with values = 0,1,2,3. For every value of . there is a separate notation to indicate subshell. For example.
| Quantum No. Subshell Notation =s P d f g____ h.... |
The first four notations :s,p,d and f are adjectives used earlier to identify spectral lines; sharp , principal, diffuse and fundamental but subsequent notations used for >3 proceed alphabetically : g,h.i.j. ...so on.
Knowing the value of n and finding out , we designate a subshell. For example, subshell for n=1,=0 is called is subshell and others are given in the form of a table
| n________________ |
| 2________________ {0_____________________2s _______________ 1______________________2p |
| 3 ______________ {0_____________________ 3s ________________ 1_____________________ 3p ________________ 2 _____________________ 3d |
| 4________________ {0_____________________ 4s _________________ 1_-____________________4p _________________ 2-____________________ 4d ________________ 3___-------_----------------------- 4f |
For atoms other than hydrogen, the energies of subshells within a given shell increases slightly with increasing
_____________________
But for other atoms, the energy increases with increasing
________________________
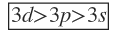
For more help in Azimuthal Quantum Number click the button below to submit your homework assignment