Theory Of Measurement Of Dipole Moment
Theory Of Measurement Of Dipole Moment Assignment Help | Theory Of Measurement Of Dipole Moment Homework Help
Theory of Measurement of Dipole Moment
The measurement or dipole moment is based upon the concept of electrical polarization of the molecules as briefly explained below:
Every molecule is made up of positively charge nuclei and negatively charge electron cloud. The centre of positive charge coincides with the centre of negative change. Hence when such a molecule is introduced between he two plates of an electric field, the positively charge nuclei and hence the centre of positive charge is attracted towards the negative plate whereas the negatively charged electron cloud is attracted towards the positive plate of the electric field. A s a result, the molecule gets distorted, as shown in . This is called polarization of the molecule ad the distorted molecule with positive and negative ends is called an electric dipole’s The molecule remains polarized so ling as the electric filed is applied butt goes back to the original state as soon as the electric field is switched off. That is why this type of polarization is called induced polarization and the electric dipole formed is called induced dipole.
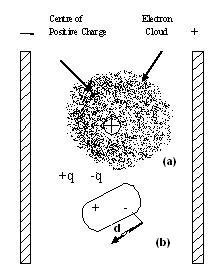
Polarization of molecule in an electric field (a)
original state (b) polarized state
Since even after polarization, the molecule is neutral as a whole, this means that the positive charge on one end must be equal to the negative change on the other. Suppose this change is q. If d is the distance of separation between the changes, then dipole moment of the induced dipole (called the induced dipole moment, represented by μ i will be given by
Evidently, the value of μ iwill depend upon the nature of the molecule and the strength of the electric field, say X We write
Where α is a constant and is called the polarizability of the given molecule.
α is found to be related to the dielectric constant, D, of the medium present between the plates i.e. the molecules being studied, according to the equation (put forward by Claudius and Menotti)
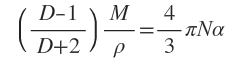
Where M is molecular mass of the molecules
P is the density of the molecules
N is Avogadro’s number.
As all quantities on the right hand side of eqn. (6.3.3) are constant ( α being constant for a given substance) independent of temperature therefore the quantity on the left hand side of eqn. (6.3.3) must also be constant, independent of temperature and depending only upon the nature of the molecules. This quantity is called induced molar polarization and is represented by P . Thus
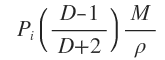
Induced molar polarization is defined as the amount of polarization produced in 1 mole of the substance when placed between the plates of an electric field of unit strength.
For more help in Theory of Measurement of Dipole Moment please click the button below to submit your homework assignment.
Every molecule is made up of positively charge nuclei and negatively charge electron cloud. The centre of positive charge coincides with the centre of negative change. Hence when such a molecule is introduced between he two plates of an electric field, the positively charge nuclei and hence the centre of positive charge is attracted towards the negative plate whereas the negatively charged electron cloud is attracted towards the positive plate of the electric field. A s a result, the molecule gets distorted, as shown in . This is called polarization of the molecule ad the distorted molecule with positive and negative ends is called an electric dipole’s The molecule remains polarized so ling as the electric filed is applied butt goes back to the original state as soon as the electric field is switched off. That is why this type of polarization is called induced polarization and the electric dipole formed is called induced dipole.
Polarization of molecule in an electric field (a)
original state (b) polarized state
Since even after polarization, the molecule is neutral as a whole, this means that the positive charge on one end must be equal to the negative change on the other. Suppose this change is q. If d is the distance of separation between the changes, then dipole moment of the induced dipole (called the induced dipole moment, represented by μ i will be given by
Evidently, the value of μ iwill depend upon the nature of the molecule and the strength of the electric field, say X We write
Where α is a constant and is called the polarizability of the given molecule.
α is found to be related to the dielectric constant, D, of the medium present between the plates i.e. the molecules being studied, according to the equation (put forward by Claudius and Menotti)
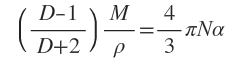
Where M is molecular mass of the molecules
P is the density of the molecules
N is Avogadro’s number.
As all quantities on the right hand side of eqn. (6.3.3) are constant ( α being constant for a given substance) independent of temperature therefore the quantity on the left hand side of eqn. (6.3.3) must also be constant, independent of temperature and depending only upon the nature of the molecules. This quantity is called induced molar polarization and is represented by P . Thus
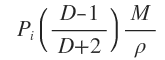
Induced molar polarization is defined as the amount of polarization produced in 1 mole of the substance when placed between the plates of an electric field of unit strength.
For more help in Theory of Measurement of Dipole Moment please click the button below to submit your homework assignment.